Animals, Agriculture & Food

Precisely Estimating Neutral Detergent Fiber Digestibilty of Biomass for Feed or Ethanol Production
WARF: P07325US
Inventors: David Combs, John Goeser
The Wisconsin Alumni Research Foundation (WARF) is seeking commercial partners interested in an in vitro method for developing precise, repeatable estimates of ruminal fiber digestion.
Overview
The amount of energy a particular type of forage contributes to a ruminant diet is a key factor in predicting animal performance. Animals fed high-energy forages gain weight faster and produce more milk.
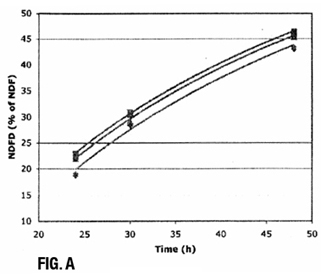
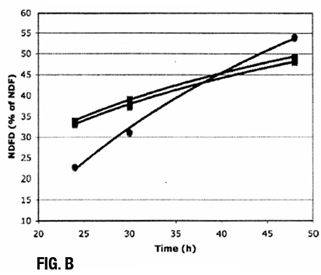
The digestibility of the fiber in a forage is a critical factor in determining energy content, but the digestibility of fiber can vary widely. Neutral detergent fiber digestibility (NDFD) is used to estimate how digestible plant fiber is in a living animal. In vitro NDFD is determined by incubating a weighed forage sample with buffers, minerals and rumen fluid from a cow. After incubation, the mixture is filtered and the undigested fiber is weighed. The difference in the weight of the fiber before and after incubation provides a measure of digestibility.
However, the ability of rumen fluid to digest fiber varies from sample to sample and from animal to animal. Additionally, the cow must be located on-site, next to where the incubation occurs. This process is too onerous and time-consuming for routine use.
Instead, the process is used to create “standardized” samples that can be used in near infrared spectroscopy (NIRS) for routine assessment of feeds of unknown digestibility. However, the variability of the rumen fluid leads to a high degree of error in the standard curve, reducing the reliability of this method and making it difficult to compare forages.
The digestibility of the fiber in a forage is a critical factor in determining energy content, but the digestibility of fiber can vary widely. Neutral detergent fiber digestibility (NDFD) is used to estimate how digestible plant fiber is in a living animal. In vitro NDFD is determined by incubating a weighed forage sample with buffers, minerals and rumen fluid from a cow. After incubation, the mixture is filtered and the undigested fiber is weighed. The difference in the weight of the fiber before and after incubation provides a measure of digestibility.
However, the ability of rumen fluid to digest fiber varies from sample to sample and from animal to animal. Additionally, the cow must be located on-site, next to where the incubation occurs. This process is too onerous and time-consuming for routine use.
Instead, the process is used to create “standardized” samples that can be used in near infrared spectroscopy (NIRS) for routine assessment of feeds of unknown digestibility. However, the variability of the rumen fluid leads to a high degree of error in the standard curve, reducing the reliability of this method and making it difficult to compare forages.
The Invention
UW-Madison researchers have developed a method for precisely estimating in vitro NDFD. This method can be used to measure fiber digestibility of biomass to assess its value in feed or ethanol production. It also can be used to calibrate a spectrophotometer or construct a more accurate and reliable standard curve for NIRS analysis.
The method involves harvesting rumen fluid from one or more animals and combining it with a carbohydrate-containing primer composition. The fluid is incubated until the rumen microbial flora reach a pre-determined, standard activity level. Then a forage sample is digested with the primed rumen fluid. As in the conventional method, the fiber content of the sample is measured before and after digestion to determine NDFD. The NDFD data then can be used to construct more reliable standards for NIRS.
The method involves harvesting rumen fluid from one or more animals and combining it with a carbohydrate-containing primer composition. The fluid is incubated until the rumen microbial flora reach a pre-determined, standard activity level. Then a forage sample is digested with the primed rumen fluid. As in the conventional method, the fiber content of the sample is measured before and after digestion to determine NDFD. The NDFD data then can be used to construct more reliable standards for NIRS.
Applications
- Assessing the digestibility of forage or other biomass to determine its value as a food source
- Assessing the value of biomass for ethanol production
- Comparing NDFD values from specific forage samples
Key Benefits
- Provides more meaningful and repeatable samples for use in routine NIRS tests on forage, feed or biofuel samples
- Because the cost of feed and animal productivity are important economic variables affected by NDFD, the ability to measure NDFD accurately and precisely may improve profitability.
- Cows may be located at a nearby facility rather than on-site.
- “Priming” procedure standardizes the rumen fluid.
- Allows the rate of digestion to be assessed over time, in addition to measuring the amount digested after a standard amount of time, to provide more meaningful predictions of energy yield
- In addition to NDFD, this method can be used to determine the digestibility of other fiber fractions, such as acid detergent fiber (ADF).
Additional Information
For More Information About the Inventors
Publications
- Goeser J.P., Hoffman P.C. and Combs D.K. 2008. An Alternative Method to Assess 24h Ruminal in Vitro Neutral Detergent Fiber Digestibility. J. Dairy Sci. 91, 35. (ABSTRACT)
Tech Fields
For current licensing status, please contact Emily Bauer at [javascript protected email address] or 608-960-9842
Figures