Medical Devices
Phospholipase Activity Provides a Simple Test for Systemic Inflammation in Acute and Chronic Disease
WARF: P02007US
Inventors: Keith Meyer, Francis Tsao
The Wisconsin Alumni Research Foundation (WARF) is seeking commercial partners interested in developing a simple, rapid and reproducible assay for systemic inflammation.
Overview
The inflammatory response plays a role in illnesses from injury to infections to allergies. Initiation of inflammation involves the activation of immune cells that trigger a cascade of events leading to phospholipase A2 (PLA2)-involved inflammatory processes.
PLA2s are enzymes that play a vital role in regulating the production of precursors to a number of pro-inflammatory lipid mediators, including prostaglandins, leukotrienes and platelet activating factor, which in turn exert a wide range of potent physiological effects. Excess production of PLA2 is associated with diseases such as systemic inflammation, allergy, brain injury, cancer development and metastasis, and cardiovascular disorders. Studying the onset and severity of disorders associated with excess PLA2 could provide useful diagnostic information as well as information on the efficacy of anti-inflammatory therapies.
PLA2s are enzymes that play a vital role in regulating the production of precursors to a number of pro-inflammatory lipid mediators, including prostaglandins, leukotrienes and platelet activating factor, which in turn exert a wide range of potent physiological effects. Excess production of PLA2 is associated with diseases such as systemic inflammation, allergy, brain injury, cancer development and metastasis, and cardiovascular disorders. Studying the onset and severity of disorders associated with excess PLA2 could provide useful diagnostic information as well as information on the efficacy of anti-inflammatory therapies.
The Invention
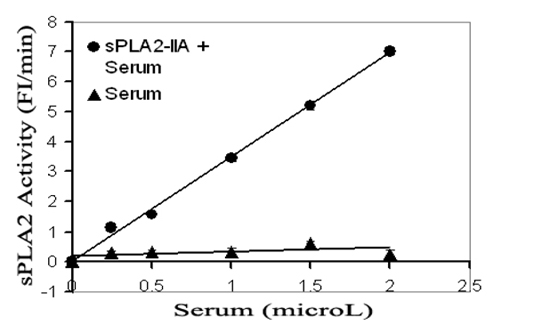
UW-Madison researchers have developed a fluorescence assay for measuring phospholipase activity, including secretory PLA2 (sPLA2) activity. This assay provides a simple, rapid and highly reproducible blood test that can be used to monitor systemic inflammation over an extended period and evaluate the effectiveness of anti-inflammatory therapies.
The assay uses a unique, fluorescently labeled liposome. When a sample containing a phospholipase is added to the liposome, the phospholipase hydrolyzes the phospholipid components of the liposome, causing a detectable change in fluorescence intensity. The degree of change indicates the activity of the phospholipase.
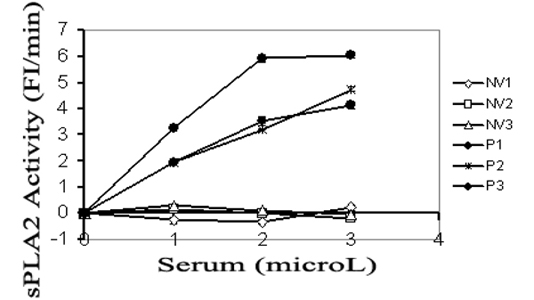
The sample can be compared to a control to determine if an individual has elevated PLA2 activity, which may indicate a disorder associated with systemic inflammation, such as sepsis, heart disease, cystic fibrosis or chronic obstructive pulmonary disease. The assay also can be used to identify agents capable of altering phospholipase activity.
Because the activity of sPLA2 in serum is a critical marker for monitoring the onset and severity of systemic inflammation in patients with sepsis or heart disease, this assay also could be used to create a portable, bedside device for early detection of hyper-inflammation. Such a device could provide accurate, real-time monitoring of changes in fluorescence intensity resulting from changes in sPLA2 activity and may enable early interventions for the prevention of multi-organ failure.
The assay uses a unique, fluorescently labeled liposome. When a sample containing a phospholipase is added to the liposome, the phospholipase hydrolyzes the phospholipid components of the liposome, causing a detectable change in fluorescence intensity. The degree of change indicates the activity of the phospholipase.
The sample can be compared to a control to determine if an individual has elevated PLA2 activity, which may indicate a disorder associated with systemic inflammation, such as sepsis, heart disease, cystic fibrosis or chronic obstructive pulmonary disease. The assay also can be used to identify agents capable of altering phospholipase activity.
Because the activity of sPLA2 in serum is a critical marker for monitoring the onset and severity of systemic inflammation in patients with sepsis or heart disease, this assay also could be used to create a portable, bedside device for early detection of hyper-inflammation. Such a device could provide accurate, real-time monitoring of changes in fluorescence intensity resulting from changes in sPLA2 activity and may enable early interventions for the prevention of multi-organ failure.
Applications
- Diagnosis and analysis of sepsis and other disorders
- Evaluation of anti-inflammatory therapies, including dietary interventions
- Identification of phospholipase modulators
- Creation of a portable, bedside device for early detection of hyper-inflammation in patients with sepsis for the prevention of multi-organ failure
Key Benefits
- Capable of determining the level of systemic inflammation response in micro-liters of serum
- Substrate and assay standards are stable for months at -20°C.
- Simple, rapid and highly reproducible
- Capable of monitoring the inflammatory process over a time course of five to six days, rather than three to four hours as seen with traditional inflammatory markers
- Phospholipase activity can be determined based on data collected at a single time point or recorded on a continuous basis.
Additional Information
For More Information About the Inventors
Publications
- Tsao F.H., Shanmuganayagam D., Zachman D.K., Khosravi M., Folts J.D. and Meyer K.C. 2007. A Continuous Fluorescence Assay for the Determination of Calcium-Dependent Secretory Phospholipase A2 Activity in Serum. Clin. Chim. Acta. 379, 119-126.
Tech Fields
For current licensing status, please contact Jennifer Gottwald at [javascript protected email address] or 608-960-9854
Figures